|
This diffusion of the product from the surface into the gas or liquid phase is called desorption. After the formation of the product, the product molecules leave the surface of the catalyst. Then, the adsorbed reactants get converted into products on the surface of the catalyst.Ĥ. During the process, the reactant molecules get attached or adsorbed on the catalyst’s surface by chemical bonding.ģ. 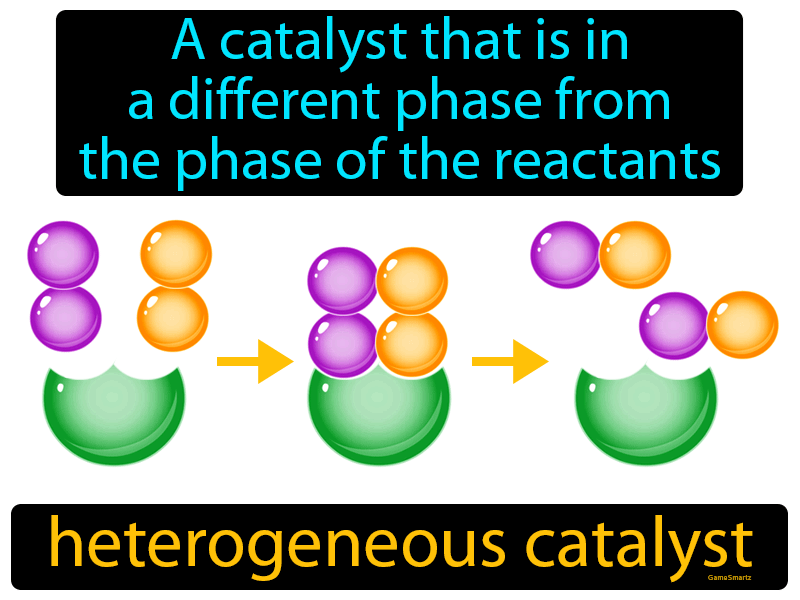
Instead, the catalytic action occurs by adsorption, where catalyst acts as adsorbent and reactant acts as adsorbate.ġ. The catalyst does not dissolve into the reacting mixture. Generally, the catalyst remains solid, while the reactants are gaseous or liquid. Unlike homogenous catalysis, the catalyst exists in a different phase than the reactant in this type of catalysis reaction. H +) yields acetic acid (CH 3COOH) and ethyl alcohol (C 2H 5OH).ĬH 3COOC 2H 5 + H 2O → CH 3COOH + C 2H 5OH 2. Therefore, the decomposition rate of ozone is higher in the presence of nitric oxide because of the catalytic activity of NO.Įxample: Hydrolysis of an ester like ethyl acetate (CH 3COOC 2H 5) in the presence of an acid catalyst (aq. As it is not consumed in the reaction, it acts as a catalyst. The nitric oxide reacts and is regenerated after ozone is decomposed. The presence of NO influences the rate of this decomposition reaction. Nitric oxide (NO) acts as a catalyst in the decomposition of ozone. Ozone is a relatively unstable molecule that decomposes to yield diatomic oxygen. Now, let us understand the process of homogeneous catalysis by citing the example of the decomposition of ozone.Īs we know, ozone is formed when oxygen molecules absorb ultraviolet light. They allow for an increase in reaction rate without an increase in temperature. These catalysts are used in various industrial applications. So, the number of collisions between reactant and catalyst is maximum. Here, the catalyst either remains uniformly dispersed in the gas phase or gets dissolved in the reactant. In this type of catalysis reaction, both catalyst and reactant remain in the same phase. For example,ĬO+3H 2 → CH 4+H 2O (when Ni is used as a catalyst)ĬO+H 2 → HCO (when Cu is used as a catalyst) It means that different products are obtained when different catalysts are used in reactions between the same reactants. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
